Home > Efficacy
Immediate initiation of JAKAVI treatment improved overall survival by 33%
- In a pooled analysis of the pivotal trials, earlier initiation of treatment with JAKAVI resulted in greater reduction in risk of death vs delayed JAKAVI initiation[2]*
OS, overall survival; RPSFT, rank-preserving structural failure time.
*After adjusting for crossover by using a RPSFT model, reduction in risk of death was greater in patients who were originally randomised to JAKAVI compared with patients who crossed over (HR [JAKAVI vs RPSFT]=0.35; 95% CI, 0.23-0.59).
In 2 pivotal trials of 528 patients, JAKAVI significantly extended survival vs BAT, including HU[3]
Pooled COMFORT-I and COMFORT-II: 5-year follow-up
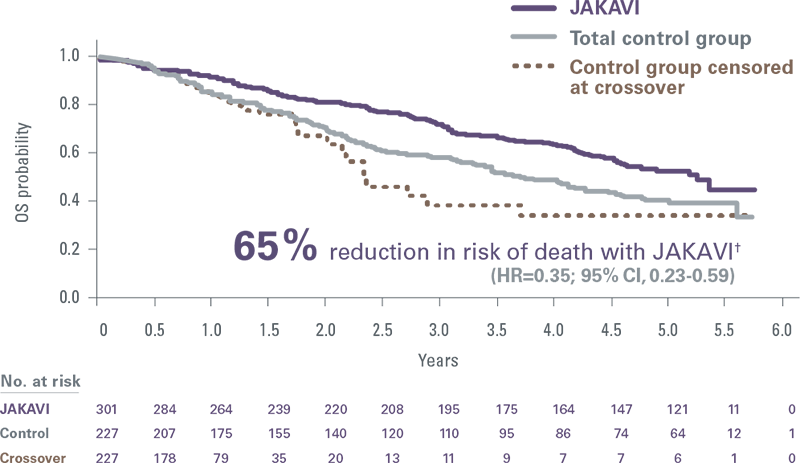
According to ELN Guidelines, the primary goal of treatment is extending overall survival for patients ineligible for stem-cell transplant[4]
COMFORT-I
- At 1-year follow-up (median: 51 weeks), JAKAVI demonstrated survival improvement (HR=0.50; 95% CI, 0.25-0.98; P=0.04)[5]
COMFORT-II
- At 3.5 years, JAKAVI treatment was associated with a 42% reduction in risk of death vs BAT (HR=0.58; 95% CI, 0.36-0.93; P=0.02)[6]
- At 5-year follow-up, there was a 33% reduction in risk of death with JAKAVI compared with BAT by ITT analysis (HR=0.67; 95% CI, 0.44-1.02; P=0.06)[1]
BAT, best available therapy; COMFORT, Controlled Myelofibrosis Study with Oral JAK Inhibitor Therapy; ELN, European LeukemiaNet; HU, hydroxyurea; ITT, intent-to-treat.
†Results from an exploratory analysis of pooled data from 528 patients in the COMFORT-I and COMFORT-II trials. Seventy percent of patients in the control group crossed over to JAKAVI during the study. The control group in COMFORT-I received placebo. The control group in COMFORT-II received BAT; the three most common were HU (47%), no medication (33%), and prednisone/prednisolone (12%). The crossover-corrected treatment effect was estimated using an RPSFT method and through censorship of survival time at the time of crossover.
Start JAKAVI earlier for superior spleen response[7]
Early intervention with JAKAVI increased the probability of spleen response by almost 60%[7]
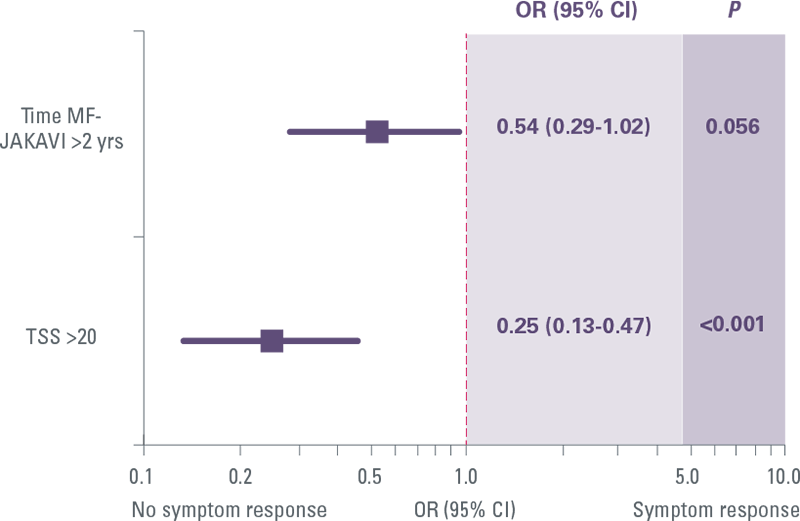
Predictors of spleen response with JAKAVI

Results from a study of 70 intermediate-1 IPSS risk MF patients treated with JAKAVI in 15 Italian (and 1 German) haematology centers. Patients received JAKAVI based on physician’s discretion after inclusion in the JUMP trial (n=51) or within a compassionate use program (n=19) and were evaluated for responses according to 2013 International Working Group-Myeloproliferative Neoplasms Research and Treatment (IWG-MRT) criteria.
Regression logistic analysis was performed to correlate spleen/symptoms responses with several baseline features, namely sex, primary/secondary MF, age >65 years, transfusion dependency, leukocytosis (>25x109/L), presence of constitutional symptoms and blast cells, palpable hepatomegaly, JAK2V617F mutation and mutation load, severe (grade 3) marrow fibrosis, Charlson Comorbidity Index ≥2, overweight, large splenomegaly (spleen length palpable ≥10 and ≥15 cm below left coast margin), TSS higher than the median value (≥20) and severely increased (≥44).
Rapid, durable spleen reduction for superior survival vs BAT (including HU)[8]
COMFORT II: Mean percentage change in spleen volume
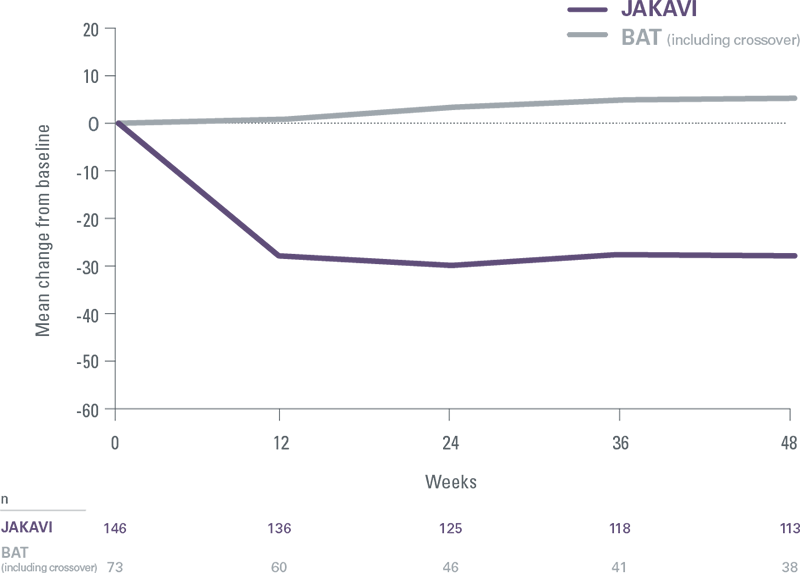
Each 10% reduction in spleen length from baseline was associated with a 9% reduction in risk of death[8]
- 97% of patients treated with JAKAVI achieved spleen reduction at any point in the study while BAT-treated patients progressively worsened[1]
- JAKAVI delivered spleen reductions as early as Week 12, with results sustained over time, regardless of JAK2V617F mutational status[9]
BAT, best available treatment.
Earlier treatment with JAKAVI increased the probability of symptom response by 54%[7]
Predictors of symptom response with JAKAVI
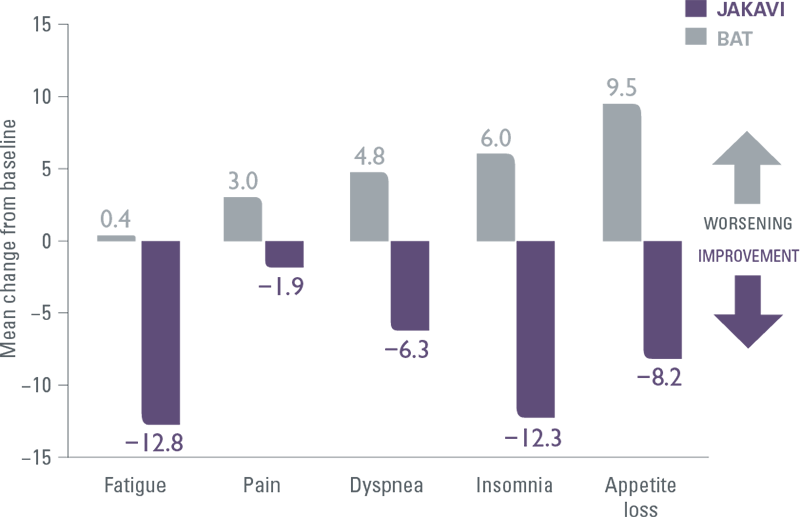
JAKAVI demonstrated superior symptom improvement compared with BAT (including HU)[9]
COMFORT-II: Mean change in EORTC QLQ-C30 symptom scores
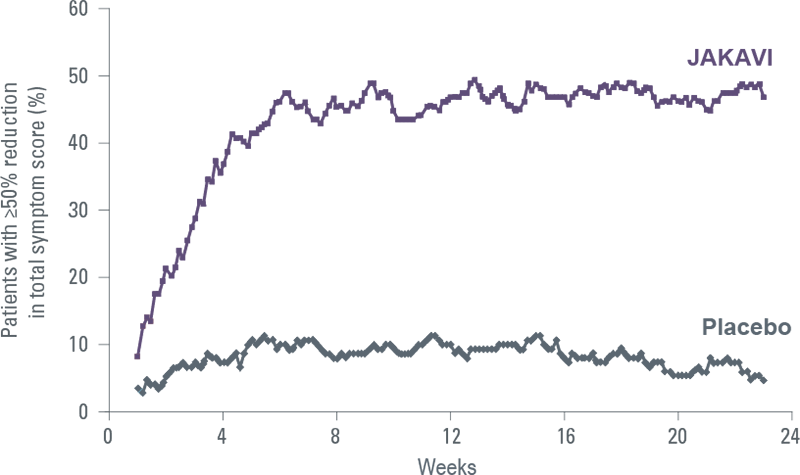
JAKAVI improves patients’ symptoms rapidly and durably[5]
COMFORT-I: Proportion of patients with ≥50% reduction in TSS over time
JAKAVI has a predictable pharmacodynamic profile with steady haemoglobin and platelet levels sustained over time
Treatment resulted in an expected and manageable decrease in haemoglobin and platelet levels, although to a lesser degree with JAKAVI vs BAT[10]
COMFORT II: Haemoglobin levels over time

COMFORT II: Platelet counts over time
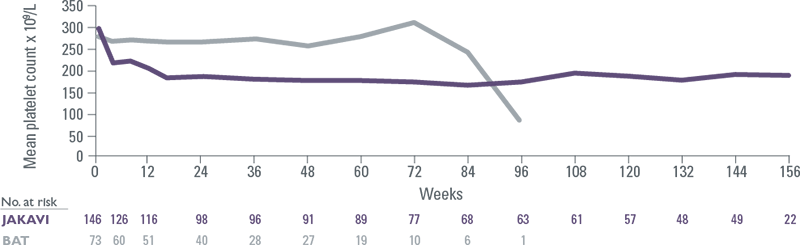
- Mean decreases in haemoglobin reached a nadir of approximately 10 g/L below baseline after 8 to 12 weeks of therapy and then gradually recovered to reach a new steady state that was approximately 5 g/L below baseline[10],[11]
- Thrombocytopenia typically presented within the first 8 weeks of therapy and was manageable with temporary dose reduction or interruption[11]
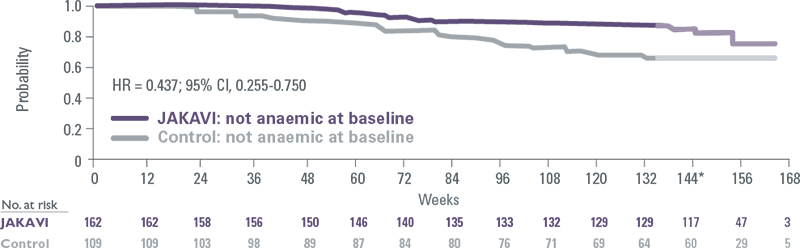
JAKAVI provided consistent overall survival benefits in patients with and without anaemia
Pooled COMFORT-I and COMFORT-II: No anaemia at baseline[12]
Pooled COMFORT-I and COMFORT-II: Anaemia at baseline[12]

- Regardless of baseline Hb levels, new or worsening anaemia that occurred during JAKAVI treatment had no effect on OS[12]
BAT, best available therapy; HU, hydroxyurea; OS, overall survival.
*After Week 144, the interpretation of the curves is affected by low numbers of patients.
References
-
Harrison CN, Vannucchi AM, Kiladjian JJ, et al. Long-term findings from COMFORT-II, a phase 3 study of ruxolitinib vs best available therapy for myelofibrosis. Leukemia. 2016;30(8):1701-1707.
-
Verstovsek S, Gotlib J, Mesa RA, et al. Long-term survival in patients treated with ruxolitinib for myelofibrosis: COMFORT-I and -II pooled analyses. J Hematol Oncol. 2017;10(1):156.
-
Verstovsek S, Gupta V, Gotlib JR, et al. A pooled overall survival (OS) analysis of 5-year data from the COMFORT-I and COMFORT-II trials of ruxolitinib for the treatment of myelofibrosis (MF). Blood. 2016;128(22):3110.
-
Barbui T, Barosi G, Birgegard G, et al. Philadelphia-negative classical myeloproliferative neoplasms: critical concepts and management recommendations from European LeukemiaNet. J Clin Oncol. 2011;29(6):761-770.
-
Verstovsek S, Mesa RA, Gotlib J, et al. Efficacy, safety and survival with ruxolitinib in patients with myelofibrosis: results of a median 3-year follow-up of COMFORT-I. Haematologica. 2013;98(12):1865-1871.
-
Harrison C, Niederwieser D, Vannuchi A, et al. Results from a 3.5 year update of COMFORT-II, a phase 3 study comparing ruxolitinib (RUX) with best available therapy (BAT) for the treatment of myelofibrosis. Abstract presented at: 19th Congress of the European Hematology Association; June 12-15, 2014; Milan, Italy.
-
Palandri F, Palumbo GA, Bonifacio M, et al. Baseline factors associated with response to ruxolitinib: an independent study on 408 patients with myelofibrosis. Oncotarget. 2017;8(45):79073-79086.
-
Vannucchi AM, Kantarjian HM, Kiladjian J-J, et al. A pooled analysis of overall survival in COMFORT-I and COMFORT-II, 2 randomized phase III trials of ruxolitinib for the treatment of myelofibrosis. Haematologica. 2015;100(9):1139-1145.
-
Harrison C, Kiladjian J-J, Al-Ali HK, et al. JAK inhibition with ruxolitinib versus best available therapy for myelofibrosis. N Engl J Med. 2012;366(9):787-798.
-
Cervantes F, Dupriez B, Pereira A, et al. New prognostic scoring system for primary myelofibrosis based on a study of the International Working Group for Myelofibrosis Research and Treatment. Blood. 2009;113(13):2895-2901.
-
JAKAVI® (ruxolitinib) tablets: EU Summary of Product Characteristics. Novartis; January 2022.
-
Gupta V, Harrison C, Hexner EO, et al. The impact of anemia on overall survival in patients with myelofibrosis treated with ruxolitinib in the COMFORT studies. Haematologica. 2016;101(12):e482-e484.
